Matching (Value 5)
|
|
|
Match each item with the correct statement below. a. | monatomic ion | f. | cation | b. | acid | g. | binary compound | c. | base | h. | anion | d. | law of definite proportions | i. | polyatomic ion | e. | law of multiple
proportions |
|
|
|
1.
|
consists of a single atom with a positive or negative charge
|
|
|
2.
|
atom or group of atoms having a negative charge
|
|
|
3.
|
atom or group of atoms having a positive charge
|
|
|
4.
|
tightly-bound group of atoms that behaves as a unit and carries a net
charge
|
|
|
5.
|
compound composed of two different elements
|
Multiple Choice (Value 24)
Identify the choice that best completes the statement or answers
the question.
|
|
|
6.
|
When Group 2A elements form ions, they ____.
a. | lose two protons | c. | lose two electrons | b. | gain two protons | d. | gain two
electrons |
|
|
|
7.
|
What is the correct name for the N 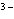 ion? a. | nitrate ion | c. | nitride ion | b. | nitrogen ion | d. | nitrite ion |
|
|
|
8.
|
When naming a transition metal ion that can have more than one common ionic
charge, the numerical value of the charge is indicated by a ____.
a. | prefix | c. | Roman numeral following the name | b. | suffix | d. | superscript after the name |
|
|
|
9.
|
Aluminum is a group 3A metal. Which ion does A1 typically form?
|
|
|
10.
|
In which of the following are the symbol and name for the ion given
correctly?
|
|
|
11.
|
The nonmetals in Groups 6A and 7A ____.
a. | lose electrons when they form ions | b. | have a numerical charge that is found by
subtracting 8 from the group number | c. | all have ions with a –1
charge | d. | end in -ate |
|
|
|
12.
|
What is the Stock name for chromic ion?
a. | chromium(I) ion | c. | chromium(III) ion | b. | chromium(II) ion | d. | chromium(IV)
ion |
|
|
|
13.
|
In which of the following are the symbol and name for the ion given
correctly?
|
|
|
14.
|
An -ate or -ite at the end of a compound name usually indicates
that the compound contains ____.
a. | fewer electrons than protons | c. | only two
elements | b. | neutral molecules | d. | a polyatomic anion |
|
|
|
15.
|
How are chemical formulas of binary ionic compounds generally written?
a. | cation on left, anion on right | b. | anion on left, cation on
right | c. | Roman numeral first, then anion, then cation | d. | subscripts first,
then ions |
|
|
|
16.
|
Which of the following is true about the composition of ionic compounds?
a. | They are composed of anions and cations. | b. | They are composed of
anions only. | c. | They are composed of cations only. | d. | They are formed from two or more nonmetallic
elements. |
|
|
|
17.
|
Which of the following formulas represents an ionic compound?
|
|
|
18.
|
In which of the following are the formula of the ionic compound and the charge
on the metal ion shown correctly?
|
|
|
19.
|
Which of the following correctly represents an ion pair and the ionic compound
the ions form?
|
|
|
20.
|
In which of the following is the name and formula given correctly?
a. | sodium oxide, NaO | c. | cobaltous chloride, CoCl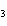 | b. | barium nitride, BaN | d. | stannic fluoride, SnF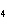 |
|
|
|
21.
|
Which of the following compounds contains the lead(II) ion?
|
|
|
22.
|
Which set of chemical name and chemical formula for the same compound is
correct?
a. | iron(II) oxide, Fe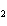 O O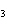 | c. | tin(IV) bromide, SnBr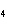 | b. | aluminum fluorate,
AlF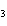 | d. | potassium chloride, K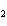 Cl Cl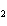 |
|
|
|
23.
|
What is the correct formula for potassium sulfite?
|
|
|
24.
|
What is the ending for the names of all binary compounds, both ionic and
molecular?
|
|
|
25.
|
Which of the following correctly shows a prefix used in naming binary molecular
compounds with its corresponding number?
a. | deca-, 7 | c. | hexa-, 8 | b. | nona-, 9 | d. | octa-, 4 |
|
|
|
26.
|
Select the correct formula for sulfur hexafluoride.
|
|
|
27.
|
What is the correct name for the compound CoCl 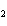 ? a. | cobalt(I) chlorate | c. | cobalt(II) chlorate | b. | cobalt(I) chloride | d. | cobalt(II)
chloride |
|
|
|
28.
|
What is the correct formula for barium chlorate?
a. | Ba(ClO)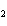 | c. | Ba(ClO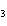 ) )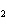 | b. | Ba(ClO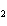 ) )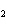 | d. | BaCl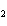 |
|
|
|
29.
|
What is the correct formula for calcium dihydrogen phosphate?
|
Numeric Response (Value 5)
|
|
|
30.
|
Write the charge of an chloride ion.
|
|
|
31.
|
What is the charge on the polyatomic ions nitrite and chlorite?
|
|
|
32.
|
What is the charge on the cation in CuSO 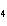 ?
|
|
|
33.
|
What is the ionic charge on the zirconium ion in the ionic compound zirconium
oxide, ZrO 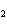 ?
|
|
|
34.
|
How many iron(II) ions combine with oxygen to form iron(II) oxide?
|
|
|
35.
|
Determine the subscript for ammonium in the chemical formula for ammonium
dichromate.
|