Matching
|
|
|
Match each item with the correct statement below.(Value 10) a. | electronegativity | f. | periodic law | b. | ionization energy | g. | cation | c. | atomic
radius | h. | period | d. | metal | i. | group | e. | transition
metal | j. | electrons |
|
|
|
1.
|
horizontal row in the periodic table
|
|
|
2.
|
vertical column in the periodic table
|
|
|
3.
|
A repetition of properties occurs when elements are arranged in order of
increasing atomic number.
|
|
|
4.
|
type of element that is a good conductor of heat and electric current
|
|
|
5.
|
type of element characterized by the presence of electrons in the d
orbital
|
|
|
6.
|
one-half the distance between the nuclei of two atoms when the atoms are
joined
|
|
|
7.
|
type of ion formed by Group 2A elements
|
|
|
8.
|
subatomic particles that are transferred to form positive and negative
ions
|
|
|
9.
|
ability of an atom to attract electrons when the atom is in a compound
|
|
|
10.
|
energy required to remove an electron from an atom
|
Multiple Choice (Value 30)
Identify the choice that best completes the statement or answers
the question.
|
|
|
11.
|
What is another name for the transition metals?
a. | noble gases | c. | Group B elements | b. | Group A elements | d. | Group C
elements |
|
|
|
12.
|
Which of the following elements is in the same period as phosphorus?
a. | carbon | c. | nitrogen | b. | magnesium | d. | oxygen |
|
|
|
13.
|
Each period in the periodic table corresponds to ____.
a. | a principal energy level | c. | an orbital | b. | an energy
sublevel | d. | a
suborbital |
|
|
|
14.
|
The modern periodic table is arranged in order of increasing atomic ____.
a. | mass | c. | number | b. | charge | d. | radius |
|
|
|
15.
|
Of the elements Pt, V, Li, and Kr, which is a nonmetal?
|
|
|
16.
|
To what category of elements does an element belong if it is a poor conductor of
electricity?
a. | transition elements | c. | nonmetals | b. | metalloids | d. | metals |
|
|
|
17.
|
In which of the following sets is the symbol of the element, the number of
protons, and the number of electrons given correctly?
a. | In, 49 protons, 49 electrons | c. | Cs, 55 protons, 132.9
electrons | b. | Zn, 30 protons, 60 electrons | d. | F, 19 protons, 19
electrons |
|
|
|
18.
|
What element has the electron configuration 1 s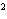 2 s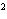 2 p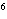 3 s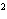 3 p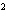 ? a. | nitrogen | c. | silicon | b. | selenium | d. | silver |
|
|
|
19.
|
Elements that are characterized by the filling of p orbitals are
classified as ____.
a. | groups 3A through 8A | c. | inner transition metals | b. | transition
metals | d. | groups 1A and
2A |
|
|
|
20.
|
Which of the following electron configurations is most likely to result in an
element that is relatively inactive?
a. | a half-filled energy sublevel | b. | a filled energy sublevel | c. | one empty and one
filled energy sublevel | d. | a filled highest occupied principal energy
level |
|
|
|
21.
|
Which subatomic particle plays the greatest part in determining the properties
of an element?
a. | proton | c. | neutron | b. | electron | d. | none of the
above |
|
|
|
22.
|
Which of the following groupings contains only representative elements?
a. | Cu, Co, Cd | c. | Al, Mg, Li | b. | Ni, Fe, Zn | d. | Hg, Cr, Ag |
|
|
|
23.
|
Which of the following is true about the electron configurations of the
representative elements?
a. | The highest occupied s and p sublevels are completely
filled. | b. | The highest occupied s and p sublevels are partially
filled. | c. | The electrons with the highest energy are in a d sublevel. | d. | The electrons with
the highest energy are in an f sublevel. |
|
|
|
24.
|
What are the Group 1A and Group 7A elements examples of?
a. | representative elements | c. | noble gases | b. | transition
elements | d. | nonmetallic
elements |
|
|
|
25.
|
Of the elements Fe, Hg, U, and Te, which is a representative element?
|
|
|
26.
|
How does atomic radius change from top to bottom in a group in the periodic
table?
a. | It tends to decrease. | c. | It first increases, then decreases. | b. | It tends to
increase. | d. | It first
decreases, then increases. |
|
|
|
27.
|
Atomic size generally ____.
a. | increases as you move from left to right across a period | b. | decreases as you
move from top to bottom within a group | c. | remains constant within a
period | d. | decreases as you move from left to right across a
period |
|
|
|
28.
|
What element in the second period has the largest atomic radius?
a. | carbon | c. | potassium | b. | lithium | d. | neon |
|
|
|
29.
|
Which of the following factors contributes to the increase in atomic size within
a group in the periodic table as the atomic number increases?
a. | more shielding of the electrons by the highest occupied energy
level | b. | an increase in size of the nucleus | c. | an increase in number of
protons | d. | fewer electrons in the highest occupied energy level |
|
|
|
30.
|
Which of the following elements has the smallest atomic radius?
a. | sulfur | c. | selenium | b. | chlorine | d. | bromine |
|
|
|
31.
|
The metals in Groups 1A, 2A, and 3A ____.
a. | gain electrons when they form ions | c. | all have ions with a 1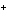 charge
charge | b. | all form ions with a negative charge | d. | lose electrons when they form
ions |
|
|
|
32.
|
Which of the following statements is NOT true about ions?
a. | Cations are positively charged ions. | b. | Anions are common among
nonmetals. | c. | Charges for ions are written as numbers followed by a plus or minus
sign. | d. | When a cation forms, more electrons are transferred to
it. |
|
|
|
33.
|
In which of the following groups of ions are the charges all shown
correctly?
|
|
|
34.
|
Which of the following elements has the smallest ionic radius?
|
|
|
35.
|
For Group 2A metals, which electron is the most difficult to remove?
a. | the first | b. | the second | c. | the
third | d. | All the electrons are equally difficult to remove. |
|
|
|
36.
|
Which of the following elements has the smallest first ionization energy?
a. | sodium | c. | potassium | b. | calcium | d. | magnesium |
|
|
|
37.
|
Compared with the electronegativities of the elements on the left side of a
period, the electronegativities of the elements on the right side of the same period tend to be
____.
a. | lower | c. | the same | b. | higher | d. | unpredictable |
|
|
|
38.
|
Which of the following decreases with increasing atomic number in Group
2A?
a. | shielding effect | c. | ionization energy | b. | ionic size | d. | number of
electrons |
|
|
|
39.
|
Which of the following factors contributes to the increase in ionization energy
from left to right across a period?
a. | an increase in the shielding effect | b. | an increase in the size of the
nucleus | c. | an increase in the number of protons | d. | fewer electrons in the highest occupied energy
level |
|
|
|
40.
|
As you move from left to right across the second period of the periodic table
____.
a. | ionization energy increases | c. | electronegativity
decreases | b. | atomic radii increase | d. | atomic mass decreases |
|
Short Answer (Answer one of the three questions below) Value 1
|
|
|
41.
|
Which group of elements in the periodic table is known as the alkali
metals?
|
|
|
42.
|
About what percent of elements is classified as metals?
|
|
|
43.
|
What is the electron configuration of sulfur?
|
Numeric Response (Answer three of the five questions below) (Value
3)
|
|
|
44.
|
How many electrons are there in the highest occupied energy level of atoms in
Group 5A elements?
|
|
|
45.
|
How many electrons are present in the d sublevel of a neutral atom of
nickel?
|
|
|
46.
|
How many electrons are in a rubidium ion (Rb 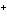 )?
|
|
|
47.
|
What is the usual charge on an ion from Group 7A?
|
|
|
48.
|
How many electrons does the ion Ca 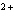 contain?
|