Matching (Value 7)
|
|
|
Match each item with the correct statement below. a. | halide ion | e. | valence electron | b. | octet rule | f. | coordination number | c. | ionic
bond | g. | metallic
bond | d. | electron dot structure |
|
|
|
1.
|
an electron in the highest occupied energy level of an atom
|
|
|
2.
|
Atoms react so as to acquire the stable electron structure of a noble
gas.
|
|
|
3.
|
a depiction of valence electrons around the symbol of an element
|
|
|
4.
|
an anion of chlorine or other halogen
|
|
|
5.
|
the force of attraction binding oppositely charged ions together
|
|
|
6.
|
the attraction of valence electrons for metal ions
|
|
|
7.
|
the number of ions of opposite charge surrounding each ion in a crystal
|
Multiple Choice (Value 28)
Identify the choice that best completes the statement or answers
the question.
|
|
|
8.
|
How many valence electrons are in an atom of phosphorus?
|
|
|
9.
|
How many valence electrons does a helium atom have?
|
|
|
10.
|
How many valence electrons are in a silicon atom?
|
|
|
11.
|
What is the name given to the electrons in the highest occupied energy level of
an atom?
a. | orbital electrons | c. | anions | b. | valence electrons | d. | cations |
|
|
|
12.
|
How does calcium obey the octet rule when reacting to form compounds?
a. | It gains electrons. | b. | It gives up electrons. | c. | It does not change
its number of electrons. | d. | Calcium does not obey the octet
rule. |
|
|
|
13.
|
What is the electron configuration of the calcium ion?
|
|
|
14.
|
The octet rule states that, in chemical compounds, atoms tend to have
____.
a. | the electron configuration of a noble gas | b. | more protons than
electrons | c. | eight electrons in their principal energy level | d. | more electrons than
protons |
|
|
|
15.
|
What is the formula of the ion formed when potassium achieves noble-gas electron
configuration?
|
|
|
16.
|
Which of the following ions has a pseudo-noble-gas electron
configuration?
|
|
|
17.
|
Which of the following elements does NOT form an ion with a charge of 1 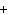 ? a. | fluorine | c. | potassium | b. | hydrogen | d. | sodium |
|
|
|
18.
|
How many electrons does nitrogen gain in order to achieve a noble-gas electron
configuration?
|
|
|
19.
|
How does oxygen obey the octet rule when reacting to form compounds?
a. | It gains electrons. | b. | It gives up electrons. | c. | It does not change
its number of electrons. | d. | Oxygen does not obey the octet
rule. |
|
|
|
20.
|
What is the electron configuration of the oxide ion (O 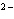 )?
|
|
|
21.
|
Which of the following occurs in an ionic bond?
a. | Oppositely charged ions attract. | b. | Two atoms share two
electrons. | c. | Two atoms share more than two electrons. | d. | Like-charged ions
attract. |
|
|
|
22.
|
A compound held together by ionic bonds is called a ____.
a. | diatomic molecule | c. | covalent molecule | b. | polar compound | d. | salt |
|
|
|
23.
|
Which of the following is true about an ionic compound?
a. | It is a salt. | c. | It is composed of anions and cations. | b. | It is held together
by ionic bonds. | d. | all of the
above |
|
|
|
24.
|
How many valence electrons are transferred from the nitrogen atom to potassium
in the formation of the compound potassium nitride?
|
|
|
25.
|
How many valence electrons are transferred from the calcium atom to iodine in
the formation of the compound calcium iodide?
|
|
|
26.
|
What is the formula unit of aluminum oxide?
|
|
|
27.
|
What is the name of the ionic compound formed from lithium and bromine?
a. | lithium bromine | c. | lithium bromium | b. | lithium bromide | d. | lithium bromate |
|
|
|
28.
|
What is the formula for sodium sulfate?
|
|
|
29.
|
Which of the following compounds has the formula KNO 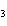 ? a. | potassium nitrate | c. | potassium nitrite | b. | potassium nitride | d. | potassium nitrogen
oxide |
|
|
|
30.
|
Which of the following pairs of elements is most likely to form an ionic
compound?
a. | magnesium and fluorine | c. | oxygen and chlorine | b. | nitrogen and sulfur | d. | sodium and
aluminum |
|
|
|
31.
|
Ionic compounds are normally in which physical state at room temperature?
a. | solid | c. | gas | b. | liquid | d. | plasma |
|
|
|
32.
|
Which of the following is NOT a characteristic of most ionic compounds?
a. | They are solids. | b. | They have low melting
points. | c. | When melted, they conduct an electric current. | d. | They are composed of
metallic and nonmetallic elements. |
|
|
|
33.
|
Which of the following particles are free to drift in metals?
a. | protons | c. | neutrons | b. | electrons | d. | cations |
|
|
|
34.
|
What is the basis of a metallic bond?
a. | the attraction of metal ions to mobile electrons | b. | the attraction
between neutral metal atoms | c. | the neutralization of protons by
electrons | d. | the attraction of oppositely charged ions |
|
|
|
35.
|
In a hexagonal close-packed crystal, every atom (except those on the surface)
has ____ neighbors.
|
Short Answer (Value 4)
|
|
|
36.
|
Give the electron configurations for mercury and its 2 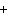 ion.
|
|
|
37.
|
Give the electron configuration for calcium the ion.
|
|
|
38.
|
Give the electron configuration for the lithium ion.
|
|
|
39.
|
Write the electron configuration diagram that shows the transfer of electrons
that takes place to form the compound sodium fluoride. Include the electron configurations of the
ions formed.
|
Numeric Response (Value 2)
|
|
|
40.
|
How many valence electrons are in bromine?
|
|
|
41.
|
How many electrons does a gallium atom give up when it becomes an ion?
|