Matching (Value 21)
|
|
|
Match each item with the correct statement below. a. | calorimeter | d. | enthalpy | b. | calorie | e. | specific heat | c. | joule | f. | heat
capacity |
|
|
|
1.
|
quantity of heat needed to raise the temperature of 1 g of water by 1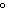 C C
|
|
|
2.
|
SI unit of energy
|
|
|
3.
|
quantity of heat needed to change the temperature of 1 g of a substance by
1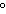 C C
|
|
|
4.
|
quantity of heat needed to change the temperature of an object by 1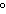 C C
|
|
|
5.
|
device used to measure the heat absorbed or released during a chemical or
physical process
|
|
|
6.
|
heat content of a system at constant pressure
|
|
|
Match each item with the correct statement below. a. | substituent | e. | asymmetric carbon | b. | structural isomers | f. | trans configuration | c. | geometric
isomers | g. | cis
configuration | d. | stereoisomers |
|
|
|
7.
|
atom or group of atoms that can take the place of a hydrogen in a parent
hydrocarbon molecule
|
|
|
8.
|
compounds that have the same molecular formula, but the atoms are joined in a
different order
|
|
|
9.
|
arrangement in which substituted groups are on the same side of a double
bond
|
|
|
10.
|
molecules in which atoms are joined in the same order but differ in the
arrangements of their atoms in space
|
|
|
11.
|
arrangement in which substituted groups are on opposite sides of a double
bond
|
|
|
12.
|
compounds that differ in the orientation of groups around a double bond
|
|
|
13.
|
carbon atom to which four different atoms or groups are attached
|
|
|
Match each item with the correct statement below. a. | aromatic compound | d. | lignite | b. | aliphatic hydrocarbon | e. | bituminous coal | c. | anthracite
coal |
|
|
|
14.
|
any straight-chain or branched-chain alkane, alkene, or alkyne
|
|
|
15.
|
any hydrocarbon compound in which a ring has bonding similar to benzene
|
|
|
16.
|
hard coal, having a carbon content of over 80%
|
|
|
17.
|
brown coal, having a carbon content of approximately 50%
|
|
|
18.
|
soft coal, having a carbon content of 70–80%
|
|
|
Match each item with the correct statement below. a. | substitution reaction | d. | hydrogenation reaction | b. | addition
reaction | e. | dehydrogenation
reaction | c. | hydration reaction |
|
|
|
19.
|
a reaction in which an atom or group of atoms replaces another atom or group of
atoms
|
|
|
20.
|
a reaction involving the addition of hydrogen to a carbon—carbon double
bond to produce an alkane
|
|
|
21.
|
a reaction involving the loss of hydrogen
|
Multiple Choice
Identify the
choice that best completes the statement or answers the question.
|
|
|
22.
|
What would likely happen if you were to touch the flask in which an endothermic
reaction were occurring?
a. | The flask would probably feel cooler than before the reaction
started. | b. | The flask would probably feel warmer than before the reaction
started. | c. | The flask would feel the same as before the reaction started. | d. | none of the
above |
|
|
|
23.
|
Which of the following is NOT a form of energy?
a. | light | c. | heat | b. | pressure | d. | electricity |
|
|
|
24.
|
When energy is changed from one form to another, ____.
a. | some of the energy is lost entirely | b. | all of the energy can be accounted
for | c. | a physical change occurs | d. | all of the energy is changed to a useful
form |
|
|
|
25.
|
If heat is released by a chemical system, an equal amount of heat will be
____.
a. | absorbed by the surroundings | c. | released by the
surroundings | b. | absorbed by the universe | d. | released by the universe |
|
|
|
26.
|
Which of the following is transferred due to a temperature difference?
a. | chemical energy | c. | electrical energy | b. | mechanical energy | d. | heat |
|
|
|
27.
|
When your body breaks down sugar completely, how much heat is released compared
to burning the same amount of sugar in a flame?
a. | The body releases more heat. | b. | The body releases less
heat. | c. | The body releases the same amount of heat. | d. | The body releases no
heat. |
|
|
|
28.
|
How many joules are in 148 calories? (1 cal = 4.18 J)
a. | 6.61 J | c. | 148 J | b. | 35.4 J | d. | 619 J |
|
|
|
29.
|
What is the specific heat of a substance if 1560 cal are required to raise the
temperature of a 312-g sample by 15 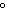 C? a. | 0.033 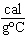 | c. | 0.99 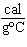 | b. | 0.33 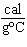 | d. | 1.33 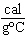 |
|
|
|
30.
|
Which of the following is a valid unit for specific heat?
|
|
|
31.
|
Which of the following has the greatest heat capacity?
a. | 1000 g of water | c. | 1 g of water | b. | 1000 g of steel | d. | 1 g of steel |
|
|
|
32.
|
Which of the following substances has the highest specific heat?
a. | steel | c. | alcohol | b. | water | d. | chloroform |
|
|
|
33.
|
By what quantity must the heat capacity of an object be divided to obtain the
specific heat of that material?
a. | its mass | c. | its temperature | b. | its volume | d. | its energy |
|
|
|
34.
|
The amount of heat transferred from an object depends on which of the
following?
a. | the specific heat of the object | c. | the mass of the
object | b. | the initial temperature of the object | d. | all of the
above |
|
|
|
35.
|
A chunk of ice whose temperature is –20 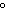 C is added to an
insulated cup filled with water at 0 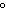 C. What happens in the cup? a. | The ice melts until it reaches the temperature of the water. | b. | The water cools
until it reaches the temperature of the ice. | c. | Some of the water freezes, so the chunk of ice
gets larger. | d. | none of the above |
|
|
|
36.
|
How many covalent bonds can each carbon atom form?
|
|
|
37.
|
How many double covalent bonds are in an alkane?
|
|
|
38.
|
What is the name of the alkane having five carbons?
a. | propane | c. | octane | b. | methane | d. | pentane |
|
|
|
39.
|
What is the simplest straight-chain alkane?
a. | graphite | c. | methane | b. | ammonia | d. | ethane |
|
|
|
40.
|
The name for an alkyl group that contains two carbon atoms is ____.
a. | diphenyl | c. | dimethyl | b. | ethyl | d. | propyl |
|
|
|
41.
|
What is the physical state of the smallest alkanes at room temperature?
a. | gas | c. | solid | b. | liquid | d. | gas or liquid |
|
|
|
42.
|
What is the general formula for a straight-chain alkane?
|
|
|
43.
|
What is the increment of change in a series of straight-chain alkanes?
|
|
|
44.
|
What is the name of the compound CH 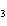 CH(CH 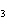 )C(CH 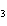 ) 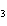 ? a. | 2,2,3-trimethylbutane | c. | 1,1,1,2-tetramethylpropane | b. | tetramethylpropane | d. | isoheptane |
|
|
|
45.
|
The condensed structural formula for 2,2,3-trimethylbutane is ____.
|
|
|
46.
|
In which of the following liquids is hexane most likely to dissolve?
a. | aqueous ammonium hydroxide | c. | rubbing alcohol | b. | vinegar | d. | octane |
|
|
|
47.
|
Which of the following compounds is an unsaturated hydrocarbon?
a. | methane | c. | nonane | b. | propyne | d. | methyl |
|
|
|
48.
|
Which of these compounds is an alkene?
a. | methane | c. | butyne | b. | nonene | d. | propanone |
|
|
|
49.
|
What is the name of the smallest alkyne?
a. | butyne | c. | methyne | b. | ethyne | d. | propyne |
|
|
|
50.
|
An organic compound that contains only carbon and hydrogen and at least one
carbon-carbon triple bond is classified as an ____.
a. | alkane | c. | alkyne | b. | alkene | d. | arene |
|
|
|
51.
|
Which of the following compounds is a structural isomer of butane?
a. | 2-methylbutane | c. | 2-methylpropane | b. | 2,2-dimethylbutane | d. | 2,2-diethylpropane |
|
|
|
52.
|
A structural isomer of hexane is ____.
a. | 2,2-dimethylbutane | c. | benzene | b. | cyclohexane | d. | 2-methylpentene |
|
|
|
53.
|
How many different atoms or groups are attached to an asymmetric carbon?
|
|
|
54.
|
In a cyclic hydrocarbon with only carbon-carbon single bonds and n number
of carbon atoms, how many hydrogen atoms are there in terms of n?
a. | 2 – n | c. | 2 + n | b. | 2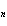 | d. | 2n |
|
|
|
55.
|
What compound is the simplest aromatic compound?
a. | methane | c. | ethyne | b. | ethene | d. | benzene |
|
|
|
56.
|
Which of the following molecules does NOT display resonance?
a. | benzene | c. | m-xylene | b. | phenylethane | d. | cyclohexane |
|
|
|
57.
|
Which of the following is NOT an important fossil fuel?
a. | petroleum | c. | natural gas | b. | hydrogen | d. | coal |
|
|
|
58.
|
Which type of coal has the highest carbon content?
a. | anthracite | c. | lignite | b. | bituminous | d. | peat |
|
|
|
59.
|
Which of the following is NOT a product obtained from the distillation of coal
tar?
a. | benzene | c. | coke | b. | phenol | d. | toluene |
|
|
|
60.
|
What is the name of the functional group in the following compound? 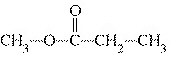 a. | halogen | c. | carbonyl | b. | ester | d. | carboxylic acid |
|
|
|
61.
|
What is the carbon skeleton of the product formed in the following reaction?
C 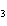 H 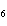 + HBr ® a. |
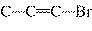 | c. |
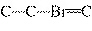 | b. |
C¾C¾C¾Br | d. |
C¾C¾Br¾C |
|
|
|
62.
|
Which of the following compounds is trichloromethane?
|
|
|
63.
|
An example of a secondary alcohol is shown by the structure ____.
|
|
|
64.
|
Which of the following compounds is a secondary alcohol?
a. |
CH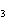 ¾CH ¾CH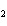 ¾CH ¾CH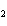 ¾CH ¾CH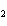 OH OH | c. |
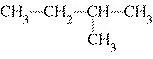 | b. |
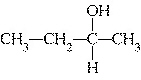 | d. |
none of the
above |
|
|
|
65.
|
What is the common name of the following alcohol? 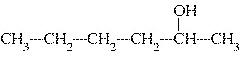 a. | sec-hexyl alcohol | c. | isohexyl
alcohol | b. | tert-hexyl alcohol | d. | hexyl alcohol |
|
|
|
66.
|
Which of the following compounds is a glycol?
|
|
|
67.
|
Which pair of formulas represents the same compound?
|
|
|
68.
|
Which of the following alcohols is used in antifreeze?
a. | ethanol | c. | ethylene glycol | b. | isopropyl alcohol | d. | glycerol |
|
|
|
69.
|
What substance is added to an organic molecule to test for the degree of
saturation?
a. | water | c. | bromine | b. | hydrogen gas | d. | hydrogen
bromide |
|
|
|
70.
|
In an addition reaction, which bond of the reactant is broken?
a. | carbon—carbon single bond | c. | carbon—carbon double
bond | b. | carbon—hydrogen single bond | d. | carbon—hydrogen double
bond |
|
|
|
71.
|
Which of the following compounds has the lowest boiling point?
a. | diethyl ether | c. | diphenyl ether | b. | 2-butanol | d. | 4-octanol |
|
|
|
72.
|
Name the compound CH 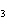 CH 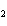 O CH 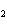 CH 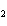 CH 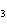 . a. | diethyl ether | c. | ethylpropyl ether | b. | dipropyl ether | d. | pentane oxide |
|
|
|
73.
|
Which carbon skeleton represents an ether?
a. |
C¾C¾C¾O¾C¾C¾C | c. |
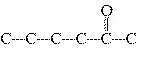 | b. |
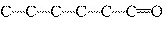 | d. |
none of the
above |
|
|
|
74.
|
What happens in a condensation reaction?
a. | head-to-tail joining of monomers | b. | side-by-side joining of
monomers | c. | cross-linking of monomers | d. | substitution of a halogen on
monomers |
|
Short Answer
|
|
|
75.
|
How much heat is required to raise the temperature of 5.5 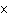
10 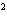 g of aluminum by 10 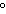 C? (specific heat of aluminum = 0.21 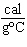 )
|
|
|
76.
|
A 55.0-g piece of copper wire is heated, and the temperature of the wire changes
from 19.0 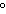 C to 86.0 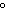 C. The amount of heat absorbed is
343 cal. What is the specific heat of copper?
|
|
|
77.
|
It takes 770 joules of energy to raise the temperature of 50.0 g of mercury by
110  C. What is the specific heat of mercury?
|
|
|
78.
|
How many carbon and hydrogen atoms are in a methane molecule?
|
|
|
79.
|
Write the general structure for halocarbon compounds.
|
|
|
80.
|
Write complete, balanced equations for the reaction of 2-pentene and water. Use
structural formulas.
|
|
|
81.
|
Write the general structure for aldehyde compounds.
|
Numeric Response
|
|
|
82.
|
How many arrangements are possible for two methyl groups with respect to a rigid
double bond?
|
|
|
83.
|
How many forms of coal are there?
|
|
|
84.
|
What percent of the composition of natural gas is methane?
|